Standardization is a linchpin of healthcare data interoperability, which allows the transformation of disparate data that describes complex concepts into a meaningful piece of information that serves as the foundation for every procedure and operation within healthcare. Governments worldwide work on defining the best practices and tools like OMOP on FHIR for interoperability and choose the standard that will simplify the collection, storage, exchange, and interpretation of health data.
Nowadays, the main focus is on the data from Electronic Health Records (EHRs) since the seamless exchange and interoperability of patient information gathered in EHRs allows for smoothing out processes from the point of view of a patient and a health worker simultaneously. However, it is not only the EHR data and interoperability that requires standardization and greatly affects the efficiency of healthcare.
One of the problems became obvious during the COVID-19 pandemic, namely the need to share research data to react proactively and effectively when facing global health threats. Today, we explore how data mapping OMOP and FHIR standards can help leverage the advantages of both data models to allow the exchange of global health and research data to support evidence-based healthcare.
HL7 FHIR: Internationally Approved Interoperability Standards
Health Level 7 International (HL7) is an ANSI-accredited non-profit health data standards developing organization aiming to create a cohesive framework for healthcare data exchange, integration, and retrieval and fosters efficient clinical practices and health service management.
Read also: TEFCA in Healthcare
FHIR (Fast Healthcare Interoperability Resources) is one of the standards developed by the HL7 organization that provides rules for exchanging healthcare data. The Ultimate Guide – What is FHIR is an excellent resource for understanding its full capabilities. Comparing FHIR vs. HL7‘s other standards, the first addresses healthcare interoperability challenges and is backed by modern web technologies such as RESTful APIs. While FHIR focuses on patient-centric data exchange, combining it with the OMOP standard unlocks advanced research and population-level insights.
By choosing FHIR-first solutions, stakeholders and developers leverage widely used open technologies and adhere to the industry requirements for the secure and standardized exchange of patient data, such as HIPAA and ONC, in the US. FHIR is a foundation for a trusted exchange of healthcare data in the US, UK, Germany, Australia, Netherlands, and more.
OMOP in Healthcare: Framework for Systematic Analysis of Disparate Databases
What does OMOP stand for? OMOP stands for the Observational Medical Outcomes Partnership that has spawned the Observational Health Data Sciences and Informatics (OHDSI) initiative that enables effective large-scale health data analytics. OHDSI spans 30 countries to support evidence-based care practices.
OMOP vs. FHIR: the Difference Between Common Data Models
The OMOP data model (CDM) standardizes disparate health-related data sources, enabling cross-institutional analytics, while FHIR focuses on real-time patient interactions. This makes OMOP healthcare ideal for population-level studies and predictive modeling.
The announcement of cooperation between the HL7 and OHDSI was an important step towards using global health and research data for improved medical research to enable personalized medicine and better outcomes. One research proves that using FHIR for OMOP data standardization minimizes information loss in transforming data into OMOP CDM, allowing seamless integration of patient genomic data.
OMOP on FHIR: The Tool for Efficient Data Management and Evidence-Based Medicine Support
The OMOP to FHIR integration – efficiently crafts predictive models for extensive patient populations, mitigating adverse events through specialized applications. Additionally, it seamlessly integrates wearable data from FHIR servers into OMOP for comprehensive population-level analysis. Let’s look at several use cases of OMOP on FHIR implementation.
Use case #1: OMOP on FHIR for the US Food and Drug Administration (FDA) Adverse Event Reporting
The challenges of the US FDA’s adverse event reporting include the complexity of handling patient data while ensuring timely and accurate reporting by incorporating the data into the HL7 Individual Case Safety Report (ICSR) in XML format in the following way:
- An asynchronous proof of concept was developed for this use case to automate XML data generation in the ICSR format.
- The next step involved integrating patient data into the OMOP database.
- The Atlas web-based tool created a cohort for this specific use case.
- The Atlas tool, supported by the API, efficiently retrieved the patient list.
- This gathered patient information was incorporated into a local database.
- Finally, the compiled data was submitted to the case manager for further processing.
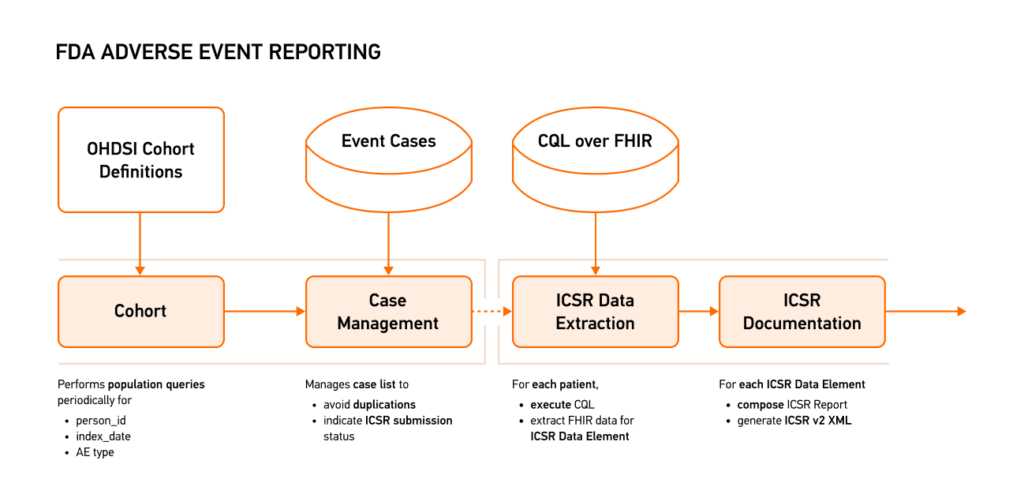
Implementing OMOP on FHIR patient data management prevented duplicates and effortlessly generated ICSR in XML format. The ICSR data extractor communicated seamlessly via API, with the case manager transmitting only new patient IDs. This approach, coupled with precise resource definition, facilitated the streamlined retrieval of adverse events from the FHIR server, enhancing the efficiency of data management and reporting.
Use Case #2: Representation of Genomic Data in HL7 FHIR and OMOP CDM
This study aimed to integrate genomic and clinical patient data for international medical research by:
- Creating semantic mapping from raw genomic data to FHIR (Fast Healthcare Interoperability Resources) and OMOP (Observational Medical Outcomes Partnership) CDM (Common Data Model)
- Standardizing and harmonizing these data sources by using the G-CDM (Genome Common Data Model) for structured representation of genomic information, development of HGNC vocabulary for consistent genetic data storage in OMOP CDM, and implementing the VCF2FHIR tool based on the General Genomic Reporting from Genomics Reporting Implementation Guide for facilitating the conversion of VCF (Variant Call Format) data into HL7 FHIR R4 format and ensuring interoperability.
Data Mapping Concept for FHIR to OMOP using G-CDM.
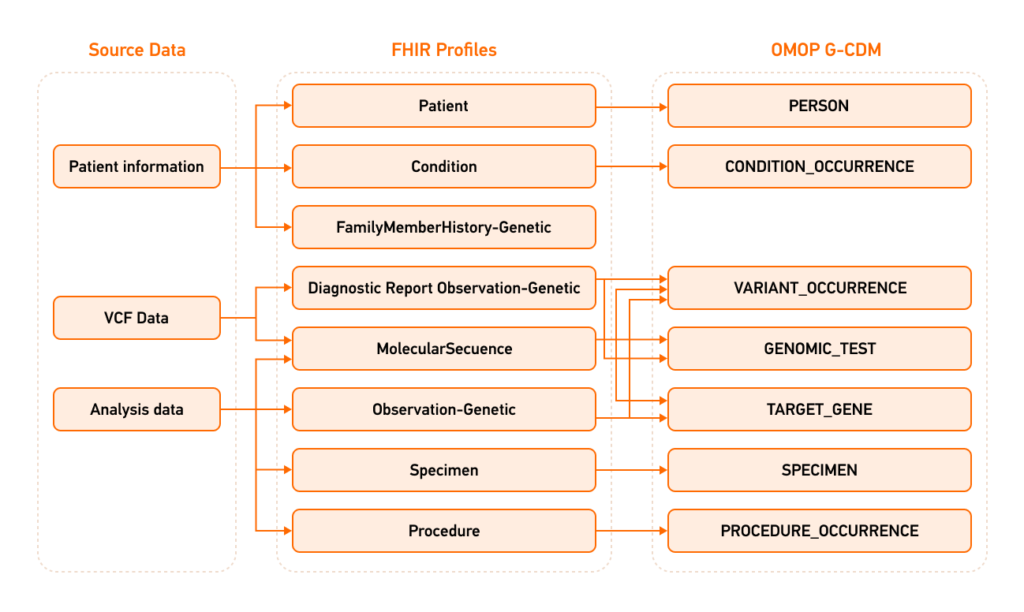
Leveraging FHIR as an intermediary between common data models has helped to achieve nearly 100% success in converting existing data to OMOP CDM, allowing researchers to analyze genomic data and use machine learning algorithms seamlessly. The study proves the importance of using OMOP and FHIR for better interoperability and efficient research and genomic data use.
Common Challenges in Mapping FHIR to OMOP
On paper, FHIR to OMOP mapping can look like a technical conversion. In real projects, it feels more like translation. FHIR is designed for exchange and clinical workflows. OMOP is designed for consistent analytics and research. The tricky part is keeping the meaning intact while you move the data.
Identifiers, de-identification, and privacy
FHIR data is rich in identifiers because care delivery depends on them. OMOP work often happens in settings where de-identification or pseudonymization is required, especially for research and multi-site studies. You still need stable linking across encounters, labs, and medications, but you cannot carry over direct identifiers. Free-text fields add another risk because they can hide sensitive details. Good mapping usually starts with a clear identity strategy, consistent pseudonymous keys, and strict rules for what gets dropped, masked, or transformed.
Status and intent elements in FHIR resources
FHIR often describes clinical activity while it is still in progress. Orders can be planned, active, completed, cancelled, or entered in error. Medications can be prescribed, dispensed, administered, or just recorded as history. OMOP prefers events that can be treated as facts in time. If you ignore status and intent, you can accidentally count planned care as delivered care. A reliable approach defines which statuses become OMOP records, which ones become metadata, and which ones should be excluded.
Data completeness and integrity
FHIR payloads are often “good enough” for exchange, but they can be thin for analytics. A lab may arrive without key context. A diagnosis may have an unclear onset. A medication may miss duration or dose details. OMOP-based studies depend on these fields for cohort logic and bias control. Integrity problems also show up when references do not resolve cleanly, or when the same clinical fact arrives multiple times with small differences. Teams usually need validation checks during mapping, plus rules for deduplication and conflict resolution.
Temporal precision in OMOP and FHIR
Time is where meaning is easy to lose. FHIR can represent multiple timestamps and time ranges, and it may include both “recorded time” and “clinical time.” OMOP needs dates that support consistent analysis, often with start and end boundaries. The hard part is choosing the right time for each domain. For labs, is it collection time or result time. For meds, is it an authored date, dispense date, or administration date. Clear rules here prevent unstable cohorts and confusing study results.
FHIR to OMOP Mapping: Methods and Tools
Most teams end up with a mix of automation and careful review. The goal is not just speed. It is repeatability, auditability, and confidence in the output.
Automated mapping tools
Automation helps with the “known knowns.” It can handle common FHIR-to-OMOP transformations, apply vocabulary lookups, and run consistent rules across multiple feeds. The best tools also log decisions, flag unknown codes, and surface exceptions so teams can review what matters most. Automation works well when it is paired with strong governance, not when it tries to hide uncertainty.
Manual curation and validation
Manual work is where the mapping becomes trustworthy. Local codes, unusual workflows, and edge cases often need a person who understands the clinical meaning. Validation also matters more than people expect. Teams usually spot-check records, review concept mapping coverage, and run small cohort tests to confirm results make clinical sense. This is often where clinicians, data analysts, and terminology experts catch issues that look “fine” technically but are wrong in practice.
Common pitfalls in mapping
A common mistake is treating FHIR like a flat export and missing the context stored in references and resource structure. Another is over-trusting coded data when units, value sets, or local code systems vary by site. Teams also run into trouble when they skip provenance. Without clear source tracking, it becomes hard to explain why two sites produce different results from the same cohort definition. Strong mapping programs keep provenance, track mapping coverage, and build feedback loops when upstream data changes.
Future Directions and Innovations in OMOP and FHIR Interoperability
FHIR and OMOP serve different purposes, but they will keep meeting in the middle. The direction is toward faster translation, stronger semantics, and fewer one-off decisions that only exist in one project.
Emerging standards and extensions
As more programs align around clearer profiles and better terminology binding, mappings become more reusable. This does not remove the need for local handling, but it reduces surprises and makes validation more predictable. Better shared conventions also make it easier to compare results across sites.
AI and predictive analytics integration
AI can support mapping in practical ways. It can suggest concept matches for local codes, flag odd unit changes, and detect anomalies that break cohorts. The main benefit is speed in triage and cleanup. High-impact decisions still need review, especially when a mapping changes exposures, outcomes, or denominators in research.
Global harmonization efforts
Multi-country research and cross-network studies increase pressure for harmonization. Differences in coding systems and privacy rules will remain, but shared playbooks, better vocabulary crosswalks, and more consistent provenance reporting can reduce friction. Over time, the most useful progress will come from making results easier to reproduce and compare across regions and organizations.
Bridging Information Gaps with Data Mapping
Apart from being a key facilitator in addressing challenges related to legacy data formats, data mapping allows for leveraging different data formats and improving the results of healthcare data analyses.
Specifically, tools like OMOP on FHIR showcase the broader advantages of FHIR data mapping in healthcare by streamlining complex processes and fostering a standardized approach to data representation, enhancing healthcare operations’ efficiency, and promoting seamless integration and analysis.
If your project involves the transformation of diverse data sources into a unified and interoperable format, consider consulting with Edenlab’s FHIR experts. Drawing from our experience in successfully converting data from old formats to FHIR when developing Kodjin Data Mapper, our team is well-equipped to assist you in harnessing the capabilities of the FHIR data model in data standardization.